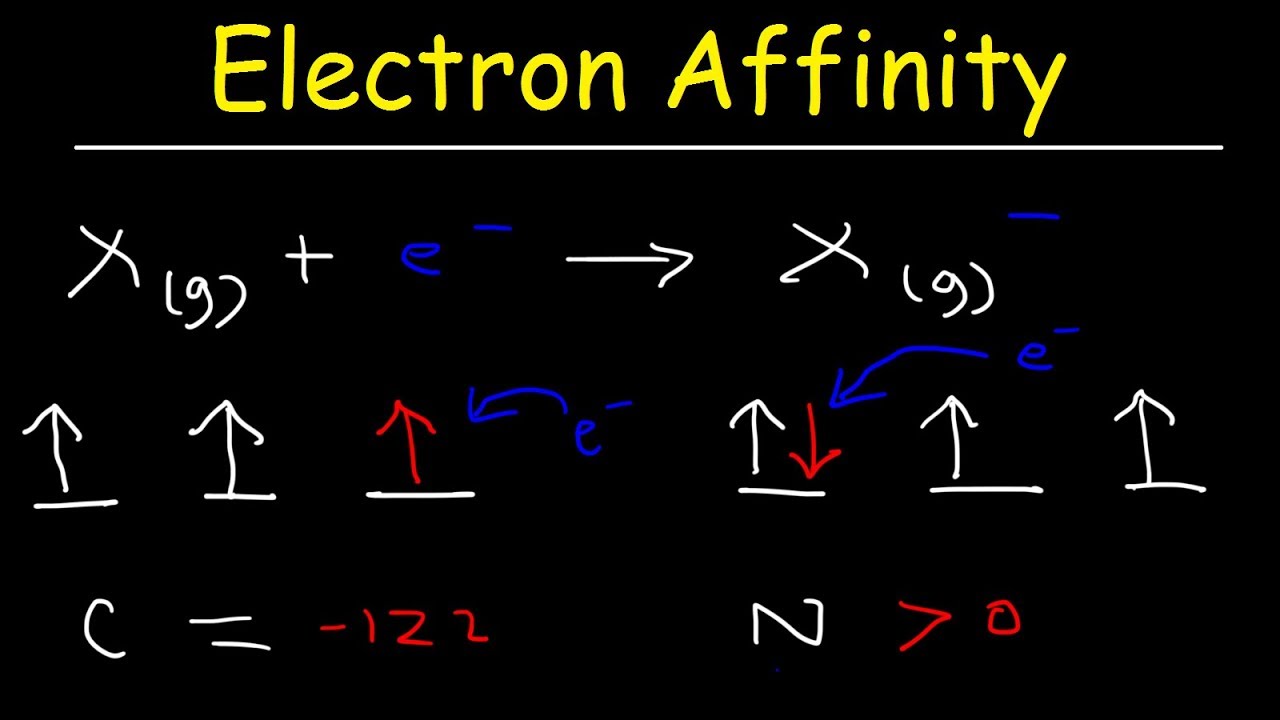
Since this electron is farther from the nucleus, it is less attracted to the nucleus and would release less energy when added. In addition, a trend of decreasing E ea going down the groups in the periodic table might be expected since the additional electron will be entering an orbital farther away from the nucleus. Typically, (i) nonmetals have more positive E ea than metals, (ii) atoms where anions are more stable than neutral atoms have a greater E ea, (iii) chlorine most strongly attracts extra electrons, (iv) mercury most weakly attracts an extra electron, (iv) E ea generally increases across a period (row) in the periodic table that is caused by the filling of the valence shell of the atom, and (v) group 17 atom releases more energy than a group 1 atom on gaining an electron because it obtains a filled valence shell and therefore is more stable. If you want to learn more about ionization energy, click here.Although E ea varies greatly across the periodic table, some patterns emerge. For this reason, they love electrons and will easily accept one from another atom to achieve it. And as you recall, these halogens only need one electron to achieve a stable electron configuration similar to that of the noble gases. Generally, electron affinity values become more negative as you move your eyes from the left of the periodic table toward the halogens (group 17). For this reason, the electron affinities of such ions are difficult to measure experimentally, because of this, we simply write their electron affinity values as being greater than zero. These positive electron affinity values mean that the negative ions of these atoms are less stable than the neutral atoms. This strong attraction makes sense because fluorine and the rest of the halogens just need a single electron to achieve an octet (8) of valence electrons similar to that of the noble gases (group 18).Īlso, on the table, you will notice that the noble gases (group 18) all have electron affinities greater than zero (positive values). And if you compare the electron affinity of fluorine to the electron affinities of the rest of the elements in group 17, you will notice that they all have a strong attraction for an electron. The large negative value shows that fluorine atom easily accepts an electron. We can write this addition as: Electron Affinity
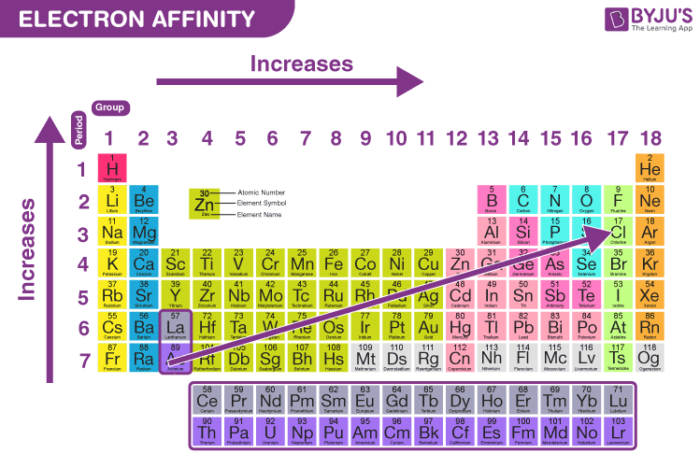
As you can see, when an electron is added to a fluorine (F) atom in the gaseous state the electron affinity is -328 kJ/mol.
